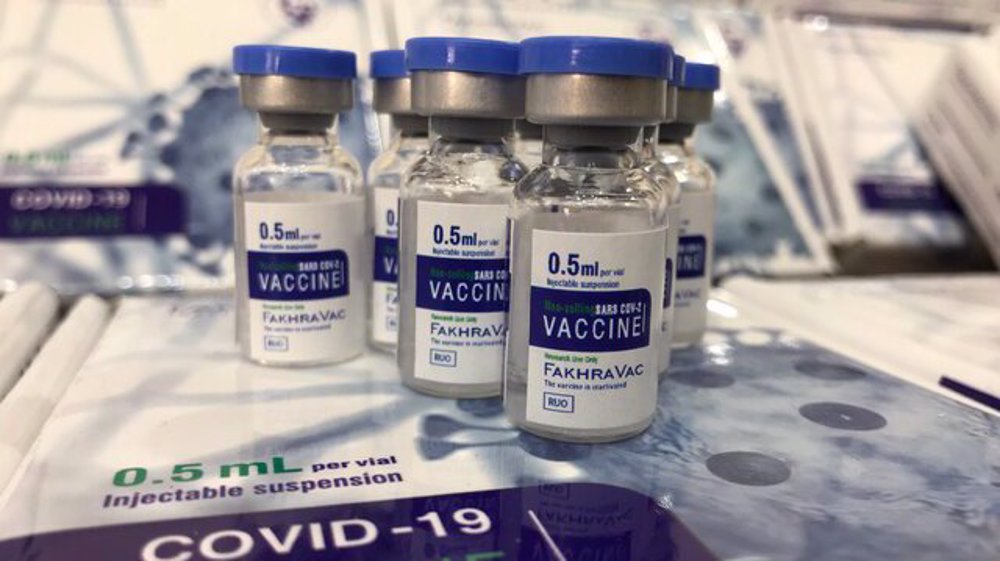
Iran to start emergency use of homegrown Fakhra COVID-19 vaccine: Defense minister
Iran’s Defense Minister Brigadier General Mohammad Reza Ashtiani says the country’s domestically-developed and produced Fakhra coronavirus vaccine has begun the third stage of clinical trial and has been authorized by the Health Ministry for emergency use.
Speaking during a visit to the production line of the vaccine on Thursday, Ashtiani added that Fakhra has successfully undergone the first and second phases of clinical trial and would feature in the country’s vaccination campaign within the next few days.
He added that the Defense Ministry is currently capable of producing about one million doses of the Fakhra vaccine on a monthly basis, emphasizing that with the finalization of the necessary infrastructure in the next three months, the production would speed up.
Iran in June launched the second phase in the human trial of Fakhra developed by the Defense Ministry’s research center formerly headed by nuclear scientist Mohsen Fakhrizadeh, who was assassinated last year.
Fakhra is an inactivated virus-based vaccine, and apparently requires two doses given by intramuscular injection 14 days apart.
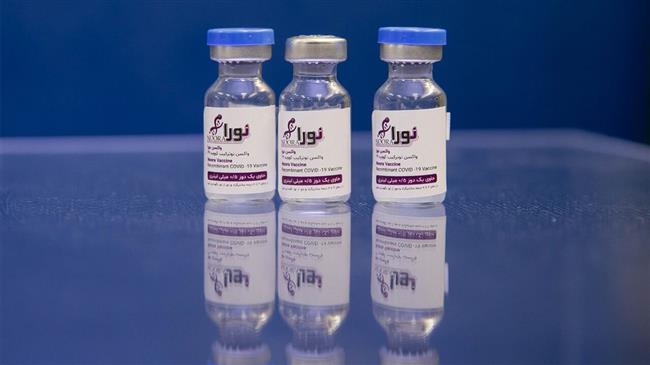
A senior official with the Islamic Revolution Guards Corps (IRGC) said on Monday that Iran’s domestically-developed and produced Noora coronavirus vaccine has also entered the second stage of human trial.
On Sunday, Iran started the third phase of the human trial of its second homegrown coronavirus vaccine, Razi Cov Pars.
Razi Cov Pars is a recombinant protein subunit vaccine containing the COVID-19 spike protein. It reportedly tutors the immune system against the virus by producing antibodies.
The vaccine includes three doses. The first two doses are said to be injectable, whilst the third dose is intranasal.
Iran has also imported foreign vaccines despite the illegal US sanctions that have seriously hampered the country’s fight against the deadly coronavirus pandemic.
Iraqi farmer killed by Israeli forces after discovering secret base: Report
Hamas slams Israeli plan to turn UNRWA compound into military bases in al-Quds
Assassination of Hamas commander sign of Israel's deceit on truce pledges: IRGC chief
Yemen reaffirms unwavering support for Palestine, Iran and Lebanon
US military presence brings ‘nothing but insecurity’ to West Asia: Qalibaf
UN rapporteur slams Israel’s systematic use of torture against Palestinian prisoners
US, Israel using ‘divisive projects’ to turn Muslims against each other: Pezeshkian
Hezbollah says Lebanon-Israel talks at ‘dead end’ as Israeli strikes continue









 This makes it easy to access the Press TV website
This makes it easy to access the Press TV website